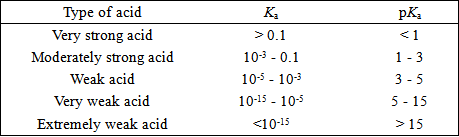
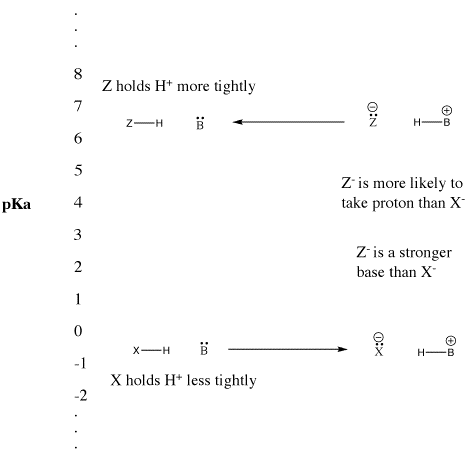
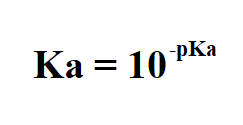SOLVED: Weak Acids Homework What is pKa if Ka 0.00001? Biochem base is a bad acid, and its pKa is usually (less or greater) than pHZ. This means that it is usually (
![PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c0a6140f88db06398f56b28ce5fd6ac92cd82d39/4-Table1-1.png)
PDF] The Influence of Ionic Strength on Apparent and Thermodynamic Parameters ( Ka , pKa ' s ) for HF and Phosphate Buffer Capacities | Semantic Scholar
![SOLVED: Consider the expressions for Ka and pKa for the acid dissociation of Ha, and fill in the blanks below Ka HA A + H+ [H+][A] Ka= [HA] pKa=-log( Ka) Remaining options: They SOLVED: Consider the expressions for Ka and pKa for the acid dissociation of Ha, and fill in the blanks below Ka HA A + H+ [H+][A] Ka= [HA] pKa=-log( Ka) Remaining options: They](https://cdn.numerade.com/ask_images/e00032db55064053b11df3a8b659b2b8.jpg)