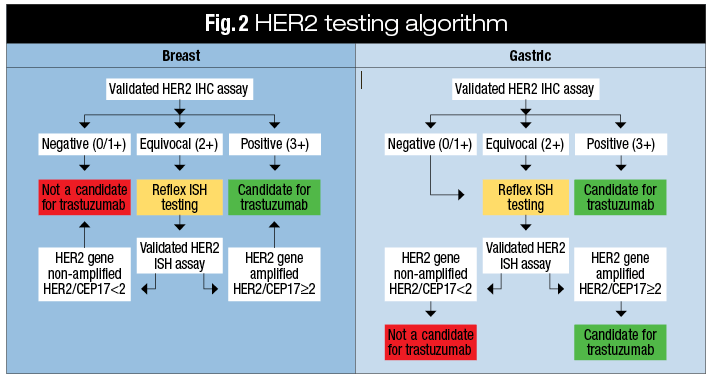
Figure 2 from Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. | Semantic Scholar
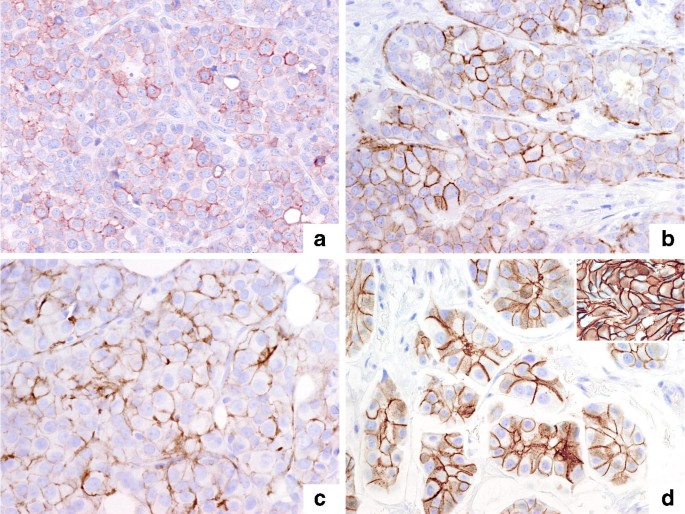
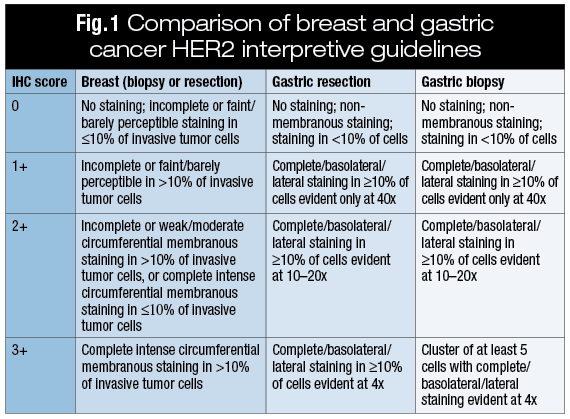
Assessment of HER2 using the 2018 ASCO/CAP guideline update for invasive breast cancer: a critical look at cases classified as HER2 2+ by immunohistochemistry | SpringerLink
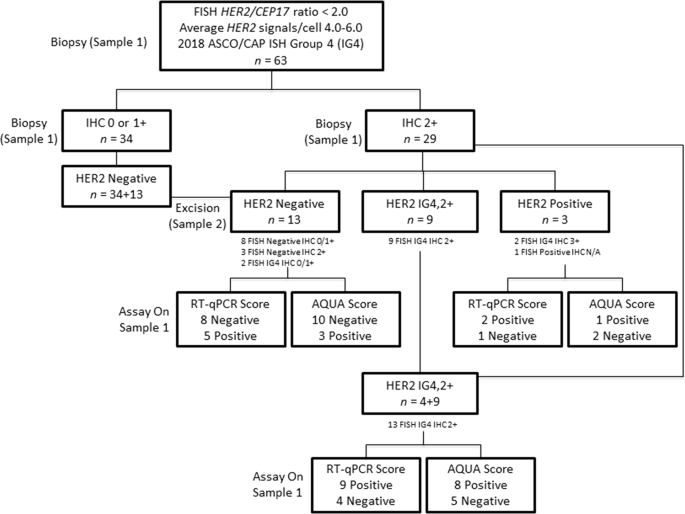
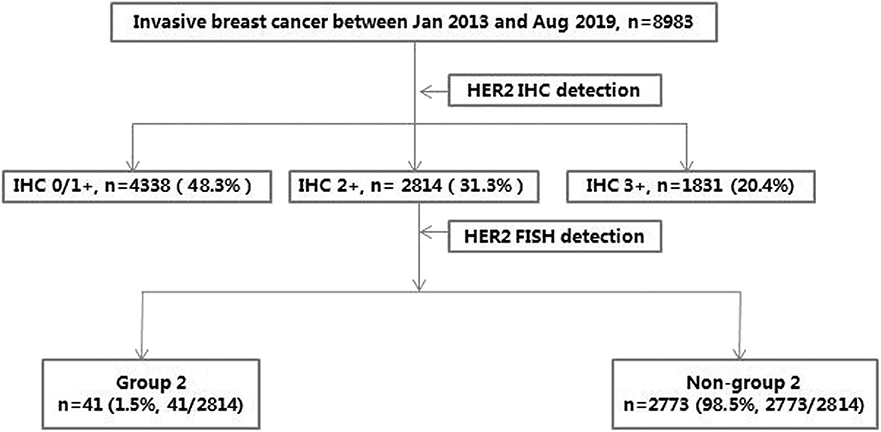
Quantitative assessments and clinical outcomes in HER2 equivocal 2018 ASCO/ CAP ISH group 4 breast cancer | npj Breast Cancer

Figure. HER2 status of breast cancer cells determined by HER2 testing;... | Download Scientific Diagram
Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pat

Figure 3 from Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. | Semantic Scholar
![PDF] THE BOTTOM LINE Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer : American Society of Clinical Oncology / College of American Pathologists Clinical Practice Guideline Focused Update Guideline Questions | Semantic Scholar PDF] THE BOTTOM LINE Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer : American Society of Clinical Oncology / College of American Pathologists Clinical Practice Guideline Focused Update Guideline Questions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/747f72db6685fa520b07391fb016bac6754d7633/9-Figure3-1.png)
PDF] THE BOTTOM LINE Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer : American Society of Clinical Oncology / College of American Pathologists Clinical Practice Guideline Focused Update Guideline Questions | Semantic Scholar

b. HER2 testing algorithm by dual probe in situ hybridisation Wolff AC... | Download Scientific Diagram
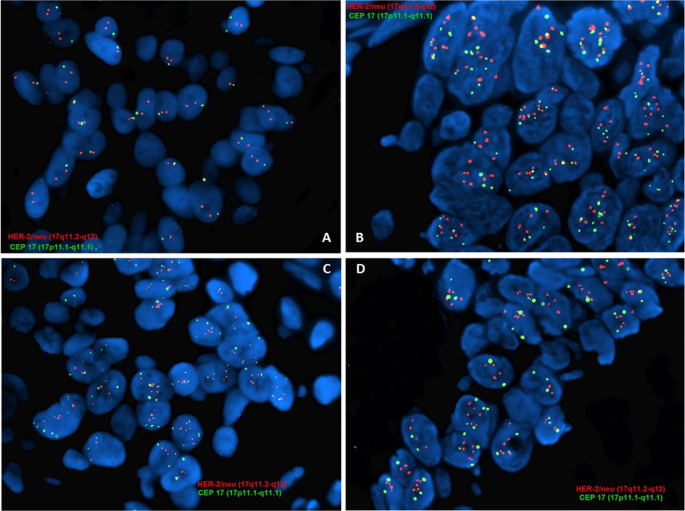
Implementation of the 2018 American Society of Clinical Oncology/College of American Pathologists Guidelines on HER2/neu Assessment by FISH in breast cancers: predicted impact in a single institutional cohort | Modern Pathology
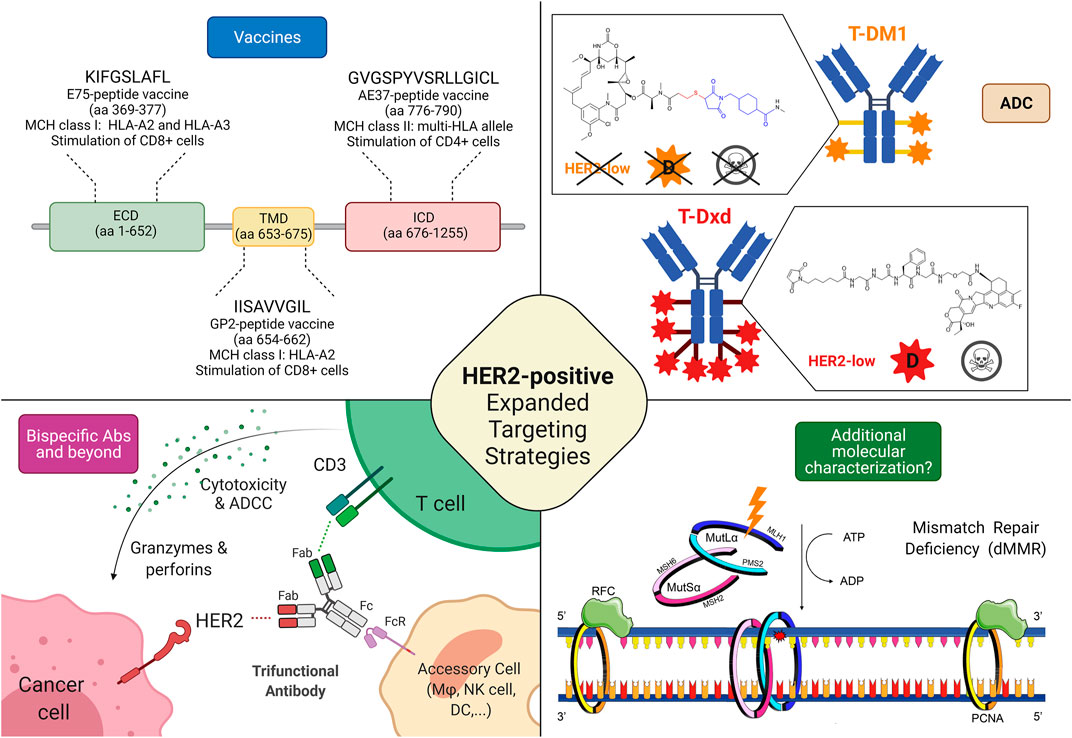
Frontiers | HER2 Low, Ultra-low, and Novel Complementary Biomarkers: Expanding the Spectrum of HER2 Positivity in Breast Cancer

a. HER2 testing algorithm by immunohistochemistry -Wolff AC et al. JCO... | Download Scientific Diagram

Performance of a HER2 testing algorithm specific for p53‐abnormal endometrial cancer - Vermij - 2021 - Histopathology - Wiley Online Library
Frontiers | Breast Cancer With a HER2 IHC2+ and FISH HER2/CEP17 Ratio ≥2.0 and an Average HER2 Gene Copy Number <4.0 per Tumor Cell: HER2 mRNA Overexpression Is a Rare Event
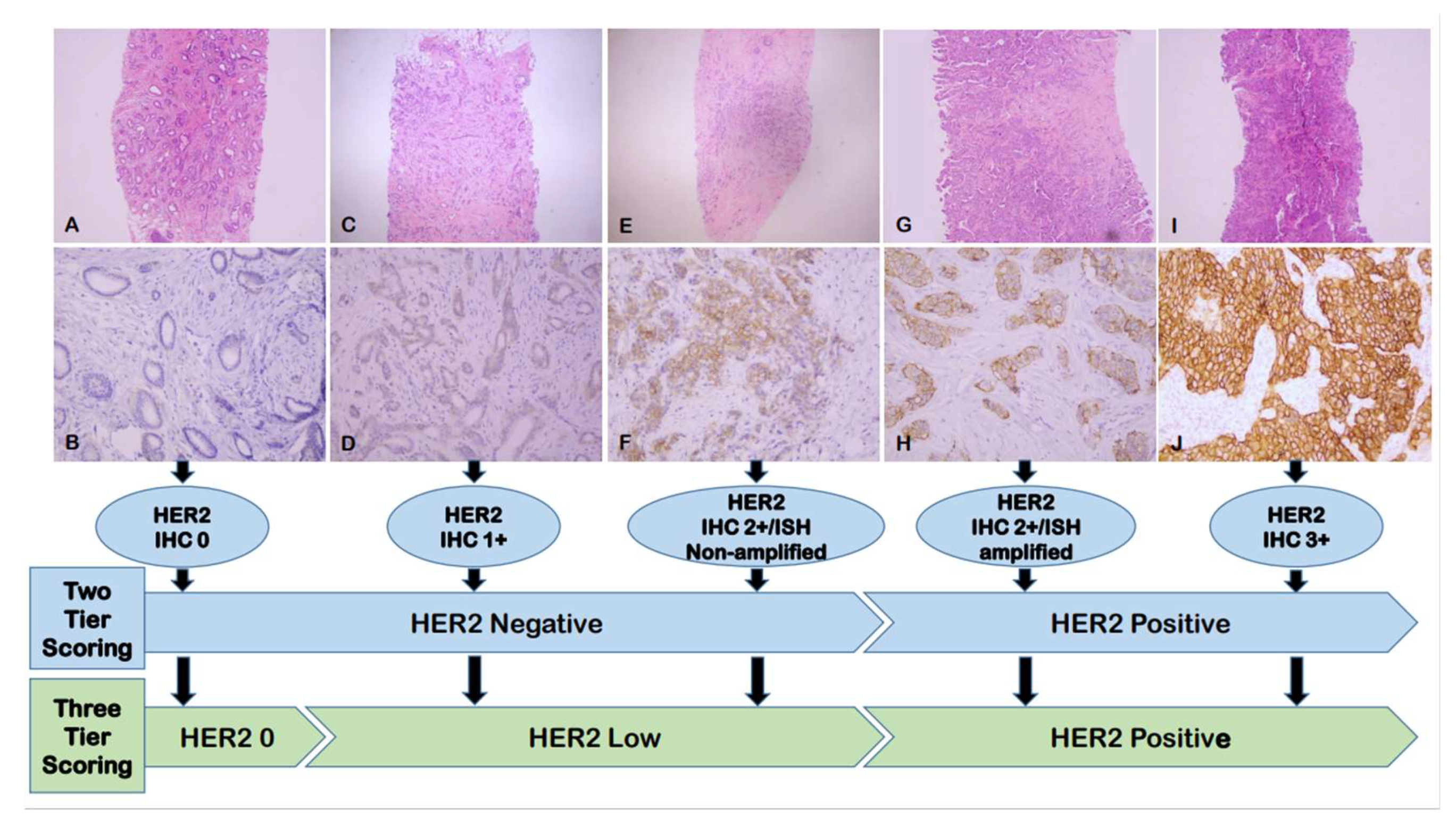
Cancers | Free Full-Text | Current Biological, Pathological and Clinical Landscape of HER2-Low Breast Cancer

ASCO/CAP 2018 breast cancer HER2 testing guidelines: summary of pertinent recommendations for practice in Australia - ScienceDirect

The differences of clinicopathologic characteristics among subgroups of reclassified HER2 fluorescence in situ hybridization (FISH) according to the ASCO/CAP 2018 breast cancer HER2 testing guidelines | Journal of Clinical Pathology
![PDF] HER2 Gene Amplification Testing by Fluorescent In Situ Hybridization (FISH): Comparison of the ASCO-College of American Pathologists Guidelines With FISH Scores Used for Enrollment in Breast Cancer International Research Group Clinical PDF] HER2 Gene Amplification Testing by Fluorescent In Situ Hybridization (FISH): Comparison of the ASCO-College of American Pathologists Guidelines With FISH Scores Used for Enrollment in Breast Cancer International Research Group Clinical](https://d3i71xaburhd42.cloudfront.net/c7289407931d6ff274350f21b4024900edadc66e/2-Figure1-1.png)
PDF] HER2 Gene Amplification Testing by Fluorescent In Situ Hybridization (FISH): Comparison of the ASCO-College of American Pathologists Guidelines With FISH Scores Used for Enrollment in Breast Cancer International Research Group Clinical