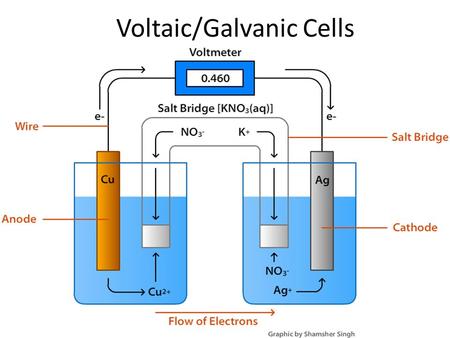
Chapter 20: Electrochemistry. © 2009, Prentice-Hall, Inc. Electrochemical Reactions In electrochemical reactions, electrons are transferred from one species. - ppt download

Three faraday of electricity is passed through molten solutions of AgNO3, NiSO4 and CrCl3 kept in three vessels using inert electrodes. The ratio in mol in which the metals Ag, Ni and

Optical analysis of Nic-Ags based nanocomposite films for promising application as ultraviolet region bandpass filters - ScienceDirect

Three faradays of electricity is passed through molten solutions of AgNO3, NiSO4 and CrCl3 kept in three vessels using inert electrodes. The ratio in mol in which the metals Ag, Ni and

SOLVED: 13.6 Post-lab Questions student constructs an electrochemical cell composed of the system: Ni(s) NiSO4(1 mol L-')IlAgNO:(1 mol . L-1)Ag(s) (a) Using the Ered values from your textbook; what cell potential would

Four Faraday of electricity were passed through aqueous solutions of `AgNO_(3), NiSO_(4), FeCl_(3)` - YouTube

SOLVED: Complete and balance the following chemical equations. NaCl (a4) + AgNO: (aq) NaNOs(aq) +AgCl(s) b) MgS (aa) + NazCO; (a4) MgCO3(aq) + NazS (s) c) Ni () + HzSO4 (a4) NiSOa(s) +

Ni-Rich LiNi0.8Co0.1Mn0.1O2 Oxide Coated by Dual-Conductive Layers as High Performance Cathode Material for Lithium-Ion Batteries | ACS Applied Materials & Interfaces

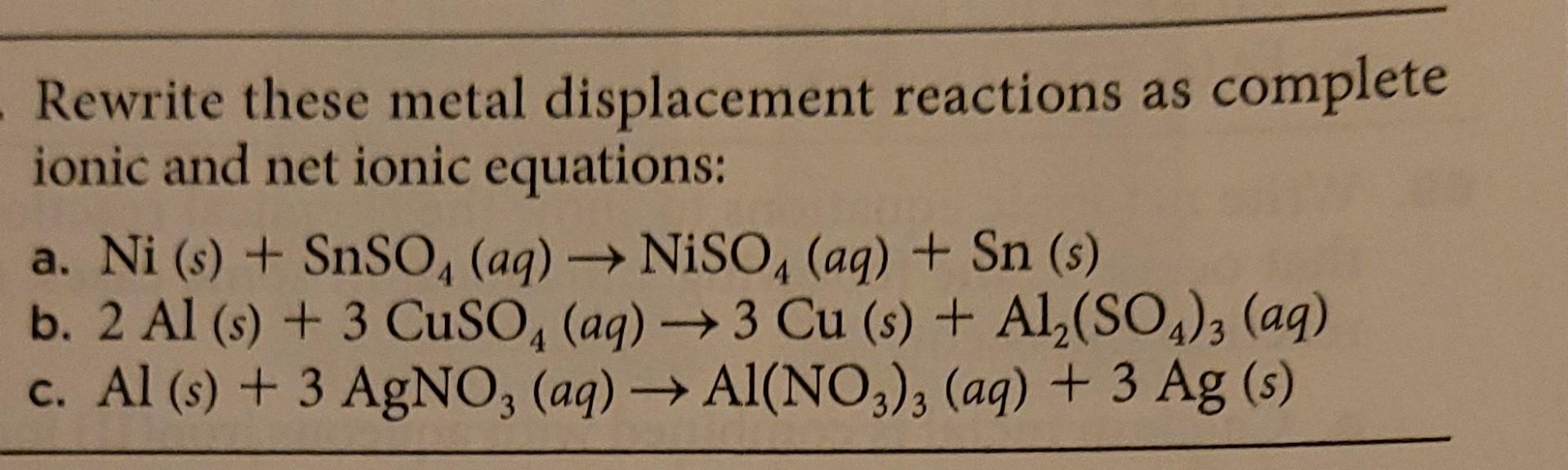

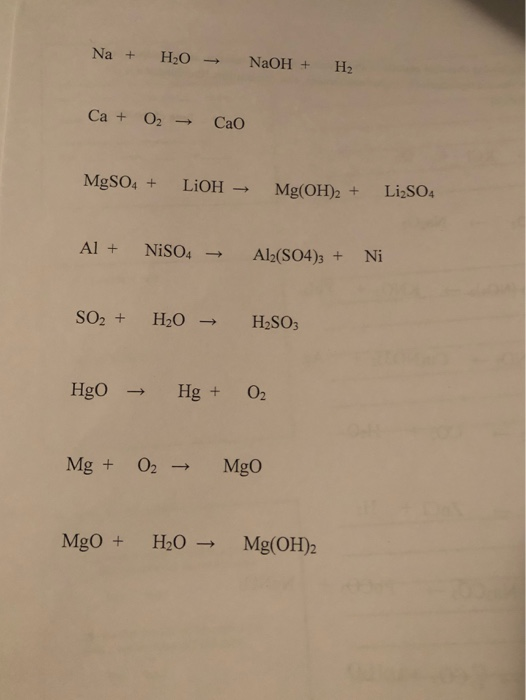








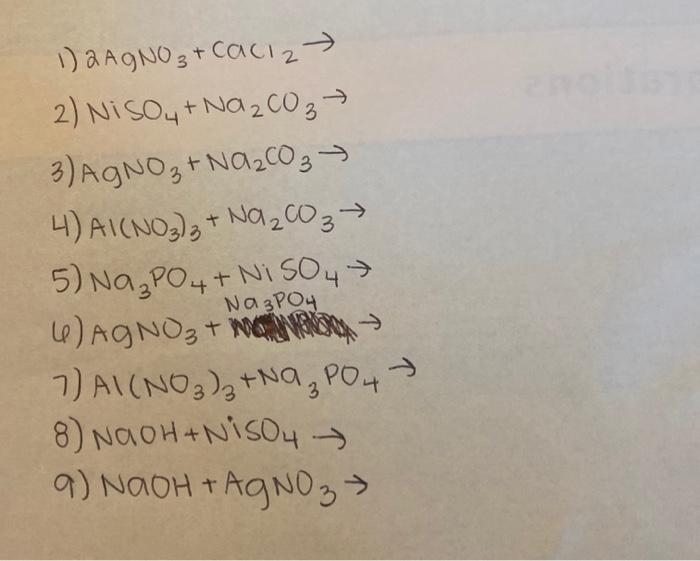



![PDF] Electrodeposition of Silver-Nickel Thin Films with a Galvanostatic Method | Semantic Scholar PDF] Electrodeposition of Silver-Nickel Thin Films with a Galvanostatic Method | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8b33b24be429a52c3e54f8d5713d82bd7bcac984/2-Figure3-1.png)